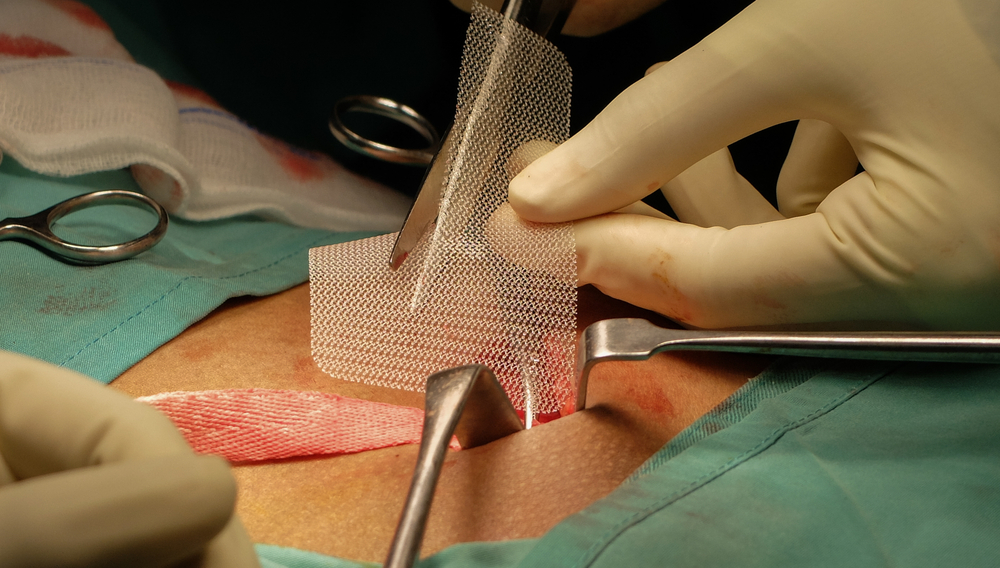
Also known as a vaginal mesh implant, a transvaginal mesh is a surgical tool used to treat stress urinary incontinence (SUI) or pelvic organ prolapse (POP). The mesh must be placed transvaginally for it to support the urethra and reconstruct the weakened pelvic muscle.
The tool often resembles a net or hammock and may take around three to five hours for the implantation procedure to end. In several cases, these surgical tools may lead to serious health complications or even death.
This has been evidenced through several lawsuits filed across the US. In this article, we will discuss transvaginal mesh litigation in detail.
Unraveling a Gynecological Scandal
Vaginal mesh implants date back to the late 1990s. In the years that followed, women started complaining of severe complications such as vaginal scarring, chronic pain, and nerve damage. There were also cases of organ perforation due to mesh erosion.
In worst-case scenarios, the implants led to death. Though researchers are still trying to understand how often these complications occur, most are life-limiting. The health risks involved with the transvaginal mesh are graver than the original condition it was designed to treat.
In 2008, a public health notification was issued by the Food and Drug Administration (FDA) stating the complications involved with vaginal mesh implants. This notification made it clear that severe (life-threatening) health complications are not a rarity.
The First Lawsuits Are Filed
Due to the rising number of health risks associated with the device, a Federal litigation started against vaginal implant manufacturers. The first-ever lawsuit was filed in 2012 against C. R. Bard’s Avaulta Plus.
The product had to be taken down the shelves as Bard lost the case with a verdict of $3.6 million in settlements. The following year, a similar lawsuit was filed against Johnson & Johnson’s Ethicon unit. The company lost the legal battle in 2013 and had to pay $3.27 million in settlements.
Less than a month later, Bard ended up losing a second lawsuit in this litigation. Besides the injuries plaintiffs had to suffer, it was alleged that the mesh manufacturers were aware of the complications involved, even the most serious ones. However, they failed to disclose them to women.
Vaginal Implants Reclassification
Around 2016, the FDA issued a reclassification order, in which all vaginal mesh implants were classified as Class III instead of Class II devices. As a part of this order, mesh manufacturers were required to obtain premarket approval if they wished to continue marketing their devices.
It was an action to protect women’s health. If the device failed to show signs of safety, the manufacturer would have to stop producing the product. As a result, all manufacturers of such devices ceased marketing transvaginal mesh for posterior compartment prolapse.
Today, no such device is commercially available as stated by the agency.
Litigation Progression and Current Position
A year after the FDA’s reclassification, a major litigation along similar lines began against Johnson & Johnson in Australia. At least 700 patients were injured by the device. The company was accused of improper device testing and downplaying the risks, not only to patients but also surgeons.
The pharmaceutical giant marketed the device as easy to implant. However, it was later found to be a complex procedure that only highly experienced surgeons could perform. In November of the same year, Australia and New Zealand banned Johnson & Johnson’s vaginal mesh implant.
Over the years that followed up to 2023, four major class actions were formed, against manufacturers like Ethicon, American Medical Systems, Boston Scientific, and C.R. Bard. There were thousands of cases under each class action.
As per the 2023 transvaginal lawsuit update, 95% of the cases filed through the years (out of over 100,000) have been settled. In total, all defendants have shelved out billions to close these suits. Currently, there is no active multi-district litigation (MDL) for this lawsuit.
Settlements and Further Scope
Are transvaginal lawsuits still being accepted? Yes, but not as a part of a class-action MDL. Since so many cases have already been settled, attorneys have a clear idea of what the future might look like. According to TruLaw, plaintiffs must follow the statute of limitations for their respective state.
In most cases, the filing window is open for two to three years from the date of the complication. If the lawsuit is successful in court, plaintiffs may expect a payout between $150,000 and $450,000. However, the same depends upon important factors like the plaintiff’s age and the severity of their injuries.
Final Thoughts
Many people believe that the transvaginal mesh litigation is no longer ongoing as all cases have been closed. However, that is untrue.
Thousands of lawsuits are still being filed across Federal courts. They are being dealt with separately against the defendant as opposed to a class-action MDL.
Alongside settlements, many manufacturers are still defending their products in court. However, healthcare providers have started looking for non-mesh alternatives to treat POP and SUI.